What happens if a vendor enters your premises without proper verification? Can your business bear the compliance risk or billing exposure? In healthcare, vendor access is a control point with a direct impact on compliance, patient data security, and billing accuracy. Credentialing delays continue to put significant financial hardship on healthcare businesses in 2026. Some hospitals estimate annual losses of more than $1 million due to credentialing delays and denied claims caused by incomplete or delayed verification.
Understanding vendor credentialing requirements helps healthcare teams reduce these risks. Healthcare facilities are increasingly facing payer and regulatory pressure to verify credentials quickly and accurately. Around 76% of credentialing teams still use manual processes, which contributes to delays and disorganized documentation.
This blog discusses essential standards, screening methods, and compliance checks associated with vendor credentialing. It also emphasizes the risks of inefficient procedures and their influence on revenue cycle activities. Each section focuses on practical activities that healthcare providers, billing teams, and compliance officials can take.
Vendor Credentialing Requirements
Vendor credentialing requirements define the verification standards for allowing vendors into healthcare environments. These controls reduce compliance risk, protect patient data, and support accurate billing processes.
What Are Vendor Credentialing Requirements
Vendor credentialing requirements include validation steps that confirm a vendor is authorized and compliant before access is granted. Healthcare facilities use these steps to control access and reduce regulatory exposure.
- Identity verification using government-issued ID
- Healthcare certification and professional license validation
- Insurance coverage verification
- Background screening
- HIPAA training completion
Why Vendor Credentialing Requirements Matter
Vendor credentialing requirements directly affect compliance, billing accuracy, and operational control. Lack of proper verification creates risk in both clinical and financial workflows.
- Prevents access by excluded vendors
- Supports compliance with federal regulations
- Reduces claim denials linked to unverified vendors
- Strengthens audit readiness
Organizations also rely on credentialing for regulatory alignment.
For example, failure to perform OIG exclusion screening for vendors can result in claim rejection and penalties.
Credentialing also connects to HIPAA training for medical vendors.
Vendors handling protected health information must complete training before system or facility access.
Vendor Credentialing Checklist: Step-by-Step Guideline
A structured vendor credentialing checklist ensures consistent verification before granting vendor access. It reduces gaps in compliance and supports accurate documentation for healthcare operations.
Core Items in Vendor Credentialing Checklist
Vendor credentialing requirements should include specific documents and validations before approval. Each item confirms compliance with healthcare and regulatory standards.
1. Government-issued identification
2. Professional license or certification
3. Liability insurance coverage
4. Background check clearance
5. Properly executed healthcare contracting services and signed agreements
6. Proof of HIPAA training for medical vendors
7. Confirmation of OIG exclusion screening for vendors
8. Tax identification details
9. Facility access authorization forms
Steps to Complete the Vendor Credentialing Checklist
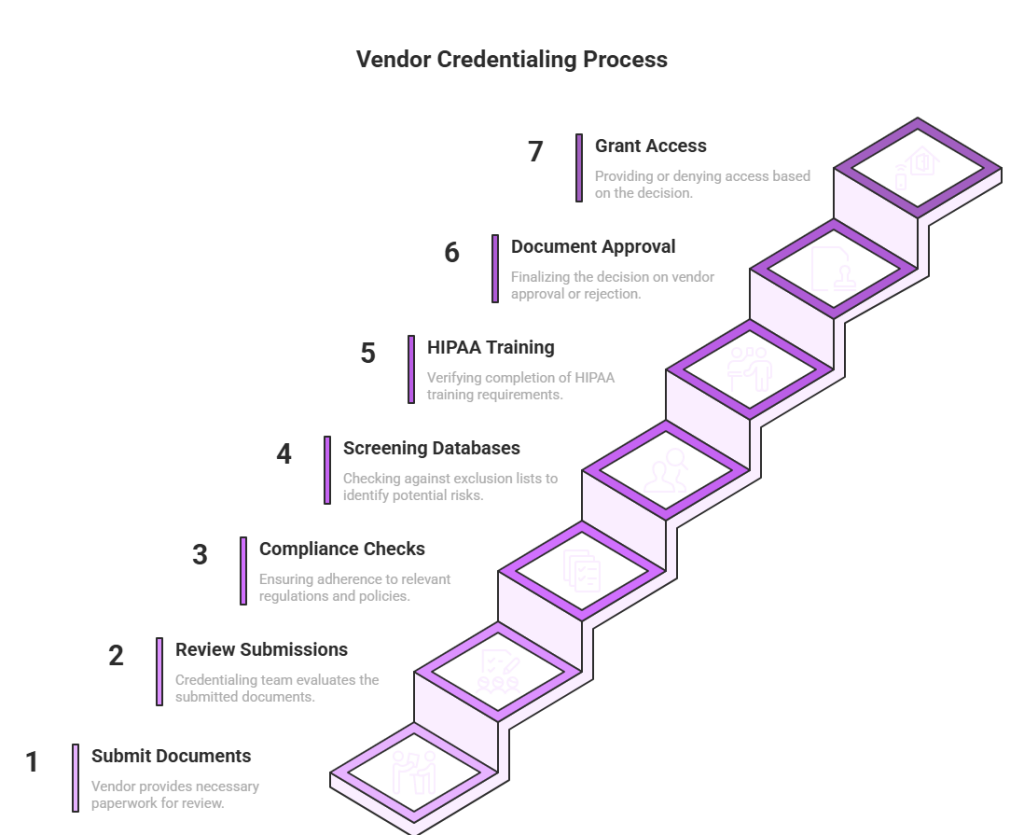
The checklist follows a structured process to ensure full verification.
1. Vendor submits required documents
2. Credentialing team reviews submissions
3. Compliance checks are performed
4. Screening against exclusion databases is completed
5. HIPAA training verification is confirmed
6. Approval or rejection is documented
7. Vendor access is granted or denied
Common Gaps in Vendor Credentialing Checklist
Many organizations face issues due to incomplete or outdated checklists. Missing expiration tracking and incomplete background checks often result in vendors operating with invalid or unverified credentials, increasing compliance risk. Delays in OIG exclusion screening for vendors can also expose organizations to claim denials and regulatory penalties.
Lack of HIPAA training for medical vendors’ verification creates gaps in data protection and increases the risk of privacy violations. Poor record documentation further limits audit readiness and makes it difficult to validate compliance during reviews or investigations.
OIG Exclusion Screening for Vendors
OIG exclusion screening for vendors is a required process to confirm vendors are not listed in federal exclusion databases. It protects healthcare organizations from compliance violations, claim denials, and financial penalties.
What Is OIG Exclusion Screening for Vendors
OIG exclusion screening for vendors involves checking vendors against the Office of Inspector General exclusion list. This list identifies individuals and entities prohibited from participating in federal healthcare programs.
- Screening against the OIG LEIE database
- Verification before onboarding
- Periodic re-screening
- Documentation of screening results
Why OIG Exclusion Screening for Vendors Is Required
This screening is mandatory for healthcare organizations participating in federal programs and is a core part of compliance controls. It prevents improper payments and reduces exposure to legal, financial, and operational risks. It also supports alignment with CMS and OIG requirements during audits and reviews.
Failure to perform OIG exclusion screening for vendors can result in claim denials, repayment obligations, and additional sanctions. Regular screening ensures only eligible vendors are approved, which protects revenue integrity and maintains compliance standards.
How to Perform OIG Exclusion Screening for Vendors
The screening process must follow a consistent and documented approach.
1. Access the OIG LEIE database
2. Enter vendor name and details
3. Verify match results carefully
4. Document screening outcome
5. Repeat screening on a set schedule
HIPAA Training for Medical Vendors
HIPAA training for medical vendors is required before vendors access protected health information (PHI) or healthcare systems. It ensures vendors understand privacy, security, and data handling standards under federal law.
What Is HIPAA Training for Medical Vendors
HIPAA training for medical vendors defines the rules for handling protected health information (PHI) within healthcare systems. It outlines requirements under the Privacy Rule and Security Rule, including proper data handling and breach response.
Training confirms that vendors understand their responsibilities before accessing patient data or systems. It is a required part of vendor credentialing requirements and must be verified for compliance.
Why HIPAA Training for Medical Vendors Is Required
HIPAA training reduces the risk of data breaches and ensures vendors follow healthcare privacy laws. It protects patient confidentiality, prevents unauthorized access, and supports compliance audits.
Failure to provide or verify training can result in penalties and regulatory action. Healthcare organizations must document vendor training before granting access to systems or facilities.
Key Components of HIPAA Training for Vendors
Effective training should cover essential compliance areas.
- Patient privacy protection
- Secure handling of electronic PHI (ePHI)
- Access control and authentication
- Incident reporting procedures
- Data breach response protocols
How to Verify HIPAA Training for Medical Vendors
Verification ensures vendors have completed the required training.
1. Collect training completion certificates
2. Verify training provider credibility
3. Confirm training content aligns with HIPAA rules
4. Record completion dates
5. Set reminders for retraining
Conclusion
Vendor credentialing requirements are essential for maintaining compliance, protecting patient data, and supporting accurate billing processes. Structured verification, including OIG exclusion screening for vendors and HIPAA training for medical vendors, reduces audit risk and prevents claim denials. Consistent credentialing practices ensure only qualified vendors have access to healthcare systems, much like the rigorous standards applied during physician credentialing services for medical staff.
Healthcare organizations that enforce standardized checklists and regular monitoring improve operational control and revenue integrity. Strong documentation and ongoing verification support audit readiness and reduce financial exposure. A well-managed credentialing process strengthens both compliance outcomes and revenue cycle performance.
FAQs
What are vendor credentialing requirements in healthcare?
Vendor credentialing requirements are the verification steps used to approve vendors before access to healthcare facilities or systems. They include identity checks, licenses, insurance, OIG screening, and HIPAA training.
How often should OIG exclusion screening for vendors be performed?
OIG exclusion screening for vendors should be done before onboarding and repeated regularly, often monthly or quarterly. Ongoing screening ensures vendors remain eligible for participation in federal healthcare programs.
Is HIPAA training for medical vendors mandatory?
Yes, HIPAA training for medical vendors is required if vendors access protected health information or systems. Healthcare organizations must verify and document training before granting access.
What happens if vendor credentialing requirements are not followed?
Failure to meet vendor credentialing requirements can lead to claim denials, audit findings, financial penalties, and compliance violations. It may also expose patient data to security risks.
How do vendor credentialing requirements impact medical billing?
Vendor credentialing requirements support accurate documentation and compliance, which helps reduce claim denials. Proper credentialing ensures only authorized vendors interact with billing and clinical systems.





