Many entrepreneurs entering the DME industry ask a direct question: “Do I really need to pay for accreditation just to sell basic medical supplies?” This concern is common because the cost structure and Medicare requirements are not clearly understood at the start. In reality, DME credentialing is not optional if you want to bill Medicare or most insurance payers. It involves enrollment, accreditation, and financial compliance rules that must all be completed before reimbursement is allowed.
In 2026, CMS continues to enforce strict requirements for DMEPOS suppliers. This includes mandatory CMS-855S enrollment, accreditation from approved bodies, and a $50,000 surety bond per NPI before billing activation.
This blog explains updated 2026 rules, including the $750 Medicare enrollment fee, accreditation requirements, exemption categories, and the financial impact of surety bonding. It also clarifies why many applications are delayed before billing ever begins.
What Is DME Credentialing and Why Does It Matter
DME Credentialing is the Medicare-required approval process that allows suppliers to bill for durable medical equipment. It is not a single registration step; it is a combination of enrollment, accreditation, and compliance validation. Without completion of all required steps, Medicare claims are not processed for payment.
This section explains the meaning of DME Credentialing and its direct impact on revenue flow. It also highlights how errors in enrollment or accreditation can stop billing access and delay reimbursement cycles.
Purpose and Scope
DME Credentialing refers to the structured approval process used by Medicare to verify suppliers before granting billing rights. It applies to suppliers of medical equipment such as oxygen systems, mobility devices, and respiratory support tools.
The scope includes:
- Medicare DME enrollment through CMS-855S form submission
- NSC supplier enrollment verification for eligibility confirmation
- Accreditation approval from CMS-recognized bodies such as ACHC or HQAA
- Compliance with DMEPOS supplier standards under Medicare rules
Why Credentialing Impacts Revenue
DME Credentialing directly controls whether a supplier can generate Medicare revenue. Billing is not activated until enrollment and accreditation are fully approved.
Key revenue impacts include:
- Suppliers cannot submit claims without active Medicare enrollment
- Errors in CMS-855S lead to claim rejections or application delays
- Accreditation gaps stop billing for specific product categories
- Delayed approval increases accounts receivable cycles
DME Credentialing vs Medicare DME Enrollment vs Accreditation
DME Credentialing is often misunderstood because three separate requirements are treated as one process. Medicare DME enrollment, accreditation, and credentialing serve different functions. Each step affects billing approval, claim acceptance, and supplier eligibility under Medicare rules.
This section separates all three components. It explains how each requirement works, where delays occur, and how errors in one step impact the entire approval process.
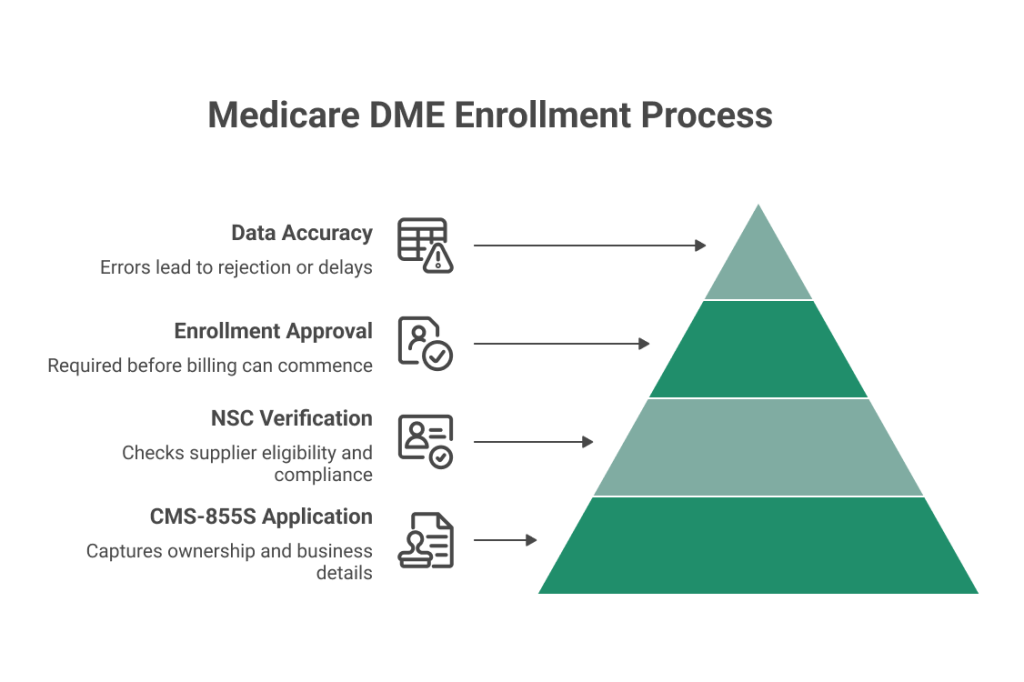
Medicare DME Enrollment (Core Requirement)
Medicare DME enrollment is the first required step for suppliers who need to bill Medicare. It is completed through the CMS-855S form and verified by the National Supplier Clearinghouse (NSC).
Key points:
- CMS-855S captures ownership, business structure, and location details
- NSC verifies supplier eligibility and compliance status
- Enrollment must be approved before billing can begin
- Errors in application data lead to rejection or extended processing time
DMEPOS Accreditation
DMEPOS accreditation confirms that a supplier meets CMS quality and operational standards. It is issued by approved accreditation bodies such as ACHC, HQAA, or The Joint Commission.
Key points:
- Required for most DME suppliers before Medicare billing approval
- Must match specific product categories being billed
- Valid for defined cycles with periodic review and renewal
- Failure to maintain accreditation leads to billing suspension
Credentialing as a Complete Process
Credentialing is the full process that combines Medicare enrollment, accreditation, and compliance validation into one operational flow. It is the stage where all requirements must align before billing activation.
Key points:
- Includes CMS-855S enrollment, accreditation, and NSC verification
- Requires consistency between business data, licensing, and accreditation scope
- Any mismatch between components delays approval or stops billing access
- Completion of all steps is required before Medicare reimbursement starts
Step-by-Step Medicare DME Enrollment Process in DME Credentialing
Medicare DME enrollment is a required process before any supplier can bill for durable medical equipment. It is completed through NSC verification and CMS-855S submission. Each step must match documentation and accreditation records.
This section breaks down the enrollment process in clear steps. It highlights where delays occur and how each stage connects to billing approval according to Medicare rules.
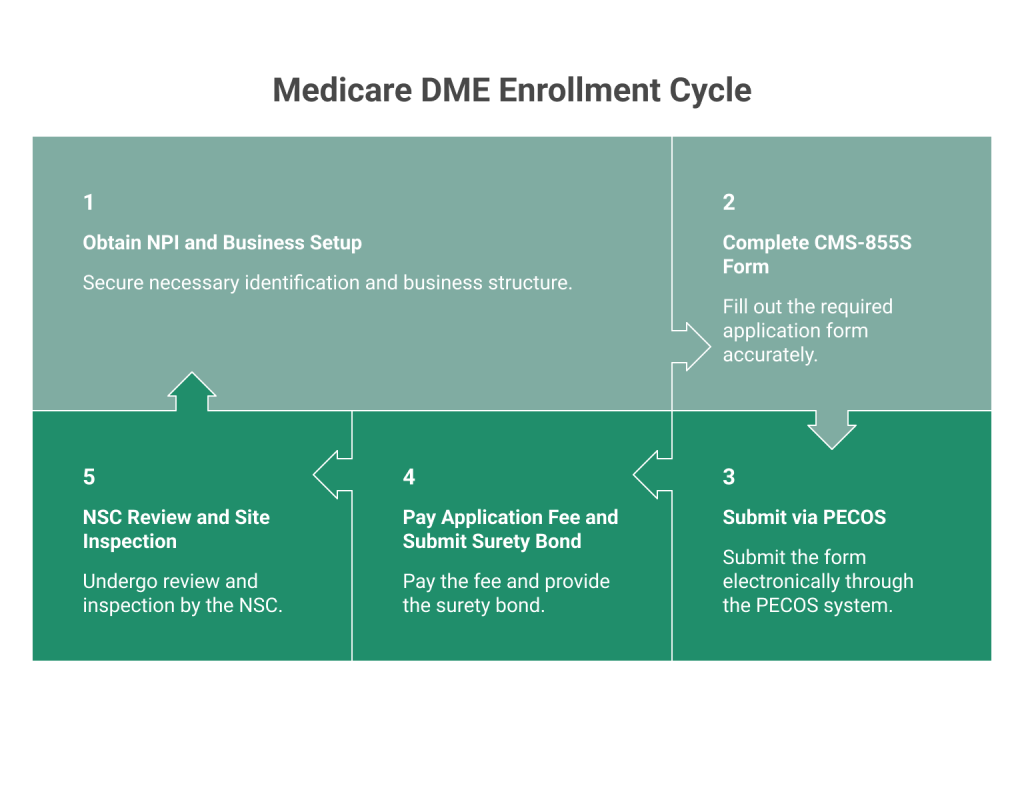
Step 1: Obtain NPI and Business Setup
The first step is to assign a National Provider Identifier (NPI) and determine the legal business structure.
- Apply for an individual or organizational NPI
- Register business entity with IRS (TIN/EIN)
- Ensure the business name matches all future enrollment records
- Confirm physical location compliance with Medicare standards
Step 2: Complete CMS-855S Form
CMS-855S is the primary Medicare enrollment application for DME suppliers.
Key requirements:
- Ownership disclosure and business structure details
- Practice location and contact information
- Accreditation details (if required)
- Product categories and supplier classification
Step 3: Submit via PECOS
PECOS is the official electronic system for Medicare enrollment submission.
- Preferred method for CMS-855S submission
- Allows document upload and status tracking
- Reduces processing delays compared to paper applications
- Requires an exact data match with supporting records
Step 4: Pay Application Fee and Submit Surety Bond
Financial requirements must be completed before enrollment approval.
- A Medicare enrollment fee is required per application cycle
- Surety bond of $50,000 per NPI is mandatory
- Bond must match ownership and enrollment details
- Missing or incorrect bond leads to an automatic delay
Step 5: NSC Review and Site Inspection
The National Supplier Clearinghouse (NSC) reviews the complete application for compliance and accuracy.
- Verification of CMS-855S details
- Review of accreditation status (if applicable)
- Site inspection for physical location compliance
- Final approval decision for Medicare billing eligibility
CMS-855S Form Explained for DME Suppliers in DME Credentialing
The CMS-855S form is the main enrollment document for DME suppliers under Medicare. It is used to collect business, ownership, and compliance information before billing approval is granted. Any mismatch in this form directly affects enrollment outcome and claim eligibility.
This section explains the key parts of the CMS-855S form and the errors that commonly delay Medicare DME enrollment. Accuracy at this stage is critical for NSC review and billing activation.
Key Sections to Complete
The CMS-855S form is divided into structured sections that must match supporting documents exactly.
Key sections include:
1. Supplier identification details
Legal business name, tax ID, and NPI must match IRS records.
2. Ownership and managing control
Disclosure of all owners, managing employees, and organizational structure.
3. Practice location information
Physical address must match site inspection records and licensing data.
4. DMEPOS product categories
Must align with accreditation scope and billed HCPCS categories.
5. Certification and compliance attestation
Confirms agreement with Medicare supplier standards and program rules.
Common Errors That Cause Delays
Most delays in Medicare DME enrollment are linked to CMS-855S form errors. These issues often result in rejection requests or extended processing time.
Common errors include:
1. Incorrect or missing NPI entries
2. Mismatch between legal business name and IRS records
3. Incomplete ownership disclosure or missing stakeholders
4. Wrong practice address compared to site inspection data
5. Product category mismatch with accreditation approval
6. Missing signatures or outdated authorization details
DMEPOS Accreditation Requirements and Exemptions in DME Credentialing
DMEPOS accreditation is a mandatory requirement for most suppliers before Medicare billing is approved. It confirms that the supplier meets CMS quality, safety, and operational standards. However, CMS allows limited exemptions under strict conditions that apply only to specific business types.
This section explains who must obtain accreditation, which entities may qualify for exemption, and where exemption decisions can create compliance risk during Medicare DME enrollment.
Who Must Obtain Accreditation
Most DME suppliers are required to complete accreditation before CMS approves billing privileges.
This includes:
- New DME suppliers enrolling for Medicare billing
- Suppliers offering oxygen, respiratory, or mobility equipment
- Providers expanding into new HCPCS-coded product categories
- Multi-location organizations operating under different NPIs
- Suppliers participating in Medicare billing for durable medical equipment services
DMEPOS Accreditation Exemptions
CMS allows only limited exemption categories, which are narrow and strictly regulated.
Common exemption cases include:
- Pharmacies with low DME revenue share (generally under CMS threshold rules)
- Suppliers with long-standing Medicare enrollment history and a clean compliance record
- Providers offering limited-scope or non-complex DME products
- Certain retail or hybrid healthcare entities with restricted Medicare billing activity
When Exemption Creates Risk
Accreditation exemption can reduce upfront cost, but it also introduces compliance exposure if not properly maintained.
Key risk situations include:
1. Growth in DME revenue exceeding exemption limits
2. Expansion into new product categories requiring accreditation
3. Ownership or structural changes affecting eligibility status
4. Incorrect classification during CMS-855S submission
5. Failure to update Medicare records after business changes
Conclusion
DME credentialing determines whether a supplier can bill Medicare and receive payment. Enrollment, accreditation, and compliance must align before claims are accepted. Errors in CMS-855S, accreditation scope, or surety bond delay approval and impact revenue.
Suppliers who follow structured Medicare DME enrollment steps reduce delays and avoid claim denials. Regular compliance review and accurate documentation protect billing access and maintain long-term reimbursement stability.
FAQs
Do all DME suppliers need accreditation to bill Medicare?
Most DME suppliers must obtain accreditation from a CMS-approved body before billing Medicare. Limited exemptions exist, but they still require full CMS-855S enrollment and NSC verification.
What is the CMS-855S form in DME credentialing?
The CMS-855S form is the official Medicare enrollment application for DME suppliers. It captures ownership, business, and compliance details required for NSC approval.
How long does Medicare DME enrollment take?
Medicare DME enrollment usually takes 30 to 90 days based on application accuracy. Delays occur due to CMS-855S errors, missing documents, or surety bond issues.
Why is a $50,000 surety bond required for DME suppliers?
CMS requires a $50,000 surety bond per NPI to protect against financial risk and fraud. Enrollment is not approved until the bond is verified and aligned with supplier records.
Can I bill Medicare before completing DME credentialing?
No, billing is not allowed until enrollment, accreditation, and NSC review are fully approved. Claims submitted before approval are denied and result in revenue loss.





