Is your clinic’s lab reimbursement about to stop completely? CMS has transitioned to a paperless system. If your CLIA data isn’t synced with payer profiles, you’re at risk. The upcoming June “Hard-Edit” deadline changes everything. After this date, the lab claims that without proper CLIA certification, payer enrollment will be denied.
This guide explains everything about CLIA certification for payer enrollment and how professional Healthcare Certification Services can help providers stay compliant. You’ll learn the CMS-116 application process from start to finish. We cover PEMS CLIA updates required for compliance. Stop your lab revenue from disappearing overnight.
Why CLIA Matters for Billing
The June deadline activates hard-edit controls. These controls automatically deny claims without a verified CLIA. There’s no grace period after June.
The Financial Impact
A clinic performing 100 lab tests weekly generates $15,000 monthly. Without proper CLIA enrollment, all $15,000 denies. Over a year, that’s $180,000 in lost revenue. The financial impact is immediate and severe. Prevention is the only option.
Who Needs CLIA Certification
Any facility testing human specimens needs CLIA. Medical offices performing rapid strep tests. Clinics are doing COVID testing. Offices running urine pregnancy tests. Urgent cares performing influenza tests. Mobile testing units. All require valid CLIA certification.
Payer Verification Systems
CMS implemented a paperless verification system. Payers now check CLIA status electronically. Medicare verifies CLIA with every claim submission. Commercial payers also require electronic verification. Missing CLIA information causes automatic denials.
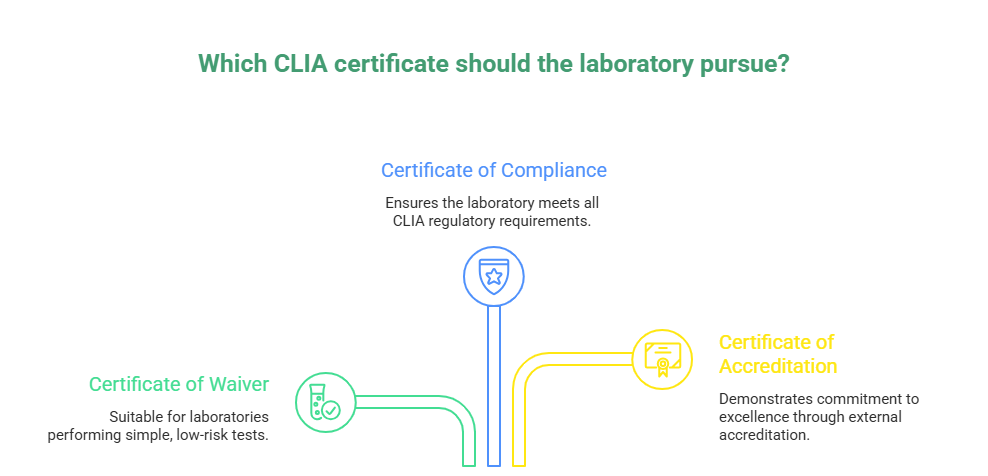
Types of CLIA Certificates
CLIA has different certificate types. Understanding these helps you get the right one.
Certificate of Waiver
The laboratory certificate of waiver covers simple tests. These tests have low error risk. They include rapid strep tests and urine pregnancy tests. COVID rapid tests qualify as waived. Blood glucose meters are waived. Most office-based testing uses waived tests.
Certificate of Compliance
This certificate allows moderate and high complexity testing. It requires more stringent oversight. Laboratory director qualifications are stricter. Quality control requirements increase. Most physician offices don’t need this level.
Certificate of Accreditation
Accredited labs receive this certificate. Accreditation comes from approved organizations. The College of American Pathologists accredits labs. This certificate allows all complexity levels. Accreditation organizations conduct regular inspections.
CLIA Certificate Types
| Certificate Type | Tests Allowed | Requirements | Typical Users |
| Certificate of Waiver | Waived tests only | Minimal | Physician offices, clinics |
| Certificate of Compliance | Moderate and high complexity | Quality systems, inspections | Small hospital labs |
| Certificate of Accreditation | All complexity levels | Accreditation organization | Large hospital labs |
CMS-116 Application Process
The CMS-116 application process is how you obtain CLIA certification. Understanding each step prevents delays.
Gather Required Information
You need your facility name and address. Provide your NPI number. Include your tax identification number. List the director of the laboratory. Specify which tests you’ll perform. Identify the certificate type needed.
Complete and Submit Form
Download form CMS-116 from the CMS website. Fill out all sections completely. Don’t leave any blanks. Sign and date the application. Include payment for the application fee. Send completed application to your state health department.
Receive Your Certificate
Processing takes 30 to 90 days, typically. The state conducts any required inspections. For waived certificates, no inspection occurs. You receive your CLIA certificate by mail. The certificate includes your unique CLIA number.
PEMS CLIA Updates
PEMS is the Provider Enrollment and Management System. PEMS CLIA updates are now mandatory for Medicare billing.
Access and Update System
Log in to the PECOS/PEMS system. Use your existing Medicare credentials. Navigate to the laboratory information section. Enter your current CLIA number. Confirm the certificate type and expiration date.
Link to NPI
Your CLIA number must link to your NPI. Each billing location needs this link. The system verifies this connection electronically. Unlinked CLIA numbers cause claim denials. Verify the link is active and correct.
Maintain Regular Updates
PEMS requires annual attestation. Update whenever CLIA information changes. The new director requires an immediate update. Certificate renewal requires a PEMS update. Regular maintenance prevents future denials.
Medicare Lab Reimbursement Requirements
Medicare lab reimbursement requirements extend beyond basic CLIA certification. Understanding these prevents unexpected denials.
Valid CLIA Certificate
Your CLIA certificate must be current and active. Expired certificates immediately stop reimbursement. The certificate must match your testing scope. Performing non-waived tests with a waiver certificate causes problems.
Proper NPI Enrollment
Your laboratory NPI must be enrolled in Medicare. Individual provider NPIs differ from facility NPIs. Use the correct NPI for lab billing. The NPI must be linked to your CLIA number. Medicare verifies this link electronically.
Compliance with Payment Policies
Medicare has specific laboratory payment policies. Some tests have frequency limitations. Others require prior authorization. Coverage varies by local Medicare contractor. Know your MAC’s specific policies.
Lab Credentialing for Clinics
Lab credentialing for clinics involves multiple payers beyond Medicare. Each has unique requirements.
Commercial Payer Requirements
Commercial payers require CLIA verification. Each payer has different processes. Some accept the CLIA number at enrollment. Others require certificate copies. Update your credentialing applications with CLIA.
Medicaid Lab Enrollment
State Medicaid programs require separate lab enrollment. CLIA certification is a prerequisite. Some states require additional licensure. Lab NPI enrollment differs from provider enrollment. Each state has unique processes and forms.
Managed Care Organizations
MCOs require separate lab credentialing. Each MCO has unique requirements. Some accept CLIA automatically. Others require additional applications. Don’t assume state Medicaid enrollment includes MCOs. Verify requirements with each plan.
Common CLIA Enrollment Mistakes
Providers make predictable CLIA enrollment errors. Avoiding these saves claim denials.
Not Obtaining CLIA Before Testing
Some clinics start testing without CLIA. They assume they can get it later. All testing requires a valid CLIA before starting. Retroactive certification doesn’t exist. Services provided without CLIA cannot be billed.
Using the Wrong Certificate Type
Performing moderate complexity tests with a waiver certificate. This creates compliance violations. It also causes claim denials. Match your certificate to the actual testing performed. Upgrade the certificate if the testing scope changes.
Forgetting to Renew
CLIA certificates expire every 2 years. Many clinics forget renewal deadlines. Expired certificates immediately stop billing. Set renewal reminders 90 days early. Don’t let certificates lapse.
CLIA Compliance Requirements
Maintaining CLIA certification requires ongoing compliance. Understanding requirements prevents certificate revocation.
Quality Control Procedures
Waived tests require quality control. Follow manufacturer instructions exactly. Document quality control results. Maintain logs of all testing. Regular quality control prevents errors.
Personnel Requirements
Certificate of waiver has minimal personnel requirements. Anyone can perform waived tests under physician direction. Higher complexity requires qualified personnel. The laboratory director must meet specific qualifications.
Record Keeping
Maintain testing logs for all tests. Keep quality control documentation. Store calibration and maintenance records. Retain records for a minimum of 2 years. Good records prove compliance.
Preparing for June Deadline
The June hard-edit deadline requires immediate action.
Verify Current CLIA Status
Pull your CLIA certificate. Check expiration date. Verify certificate type matches testing. Confirm the CLIA number is correct. Contact the state if the certificate is missing.
Update PEMS Immediately
Log in to PECOS/PEMS today. Enter your CLIA information. Verify all details are accurate. Save changes and confirm. Don’t delay this critical step.
Notify All Payors
Send CLIA information to every payer. Include Medicare, Medicaid, and commercial. Provide certificate copies if requested. Confirm receipt of updates. Document all notifications.
What Happens After June
Understanding post-deadline consequences prepares you.
Automatic Claim Denials
All lab claims without a verified CLIA denial. Denials happen at submission. No manual review occurs. The system automatically rejects claims. This affects all payers implementing hard edits.
Revenue Loss
Every denied lab claim is lost revenue. For high-volume testing clinics, losses are severe. $50,000 to $200,000 monthly. The financial impact threatens practice viability. Prevention is the only solution.
No Retroactive Fixes
You cannot fix CLIA and resubmit old claims. Timely filing limits apply. Most payers allow 90 to 180 days. CLIA fixes take time. Many claims will exceed filing limits.
Conclusion
CLIA certification for payer enrollment is mandatory for all lab billing. The June hard-edit deadline makes this critical. Complete the CMS-116 application process if you lack certification. Update PEMS CLIA information immediately. Obtain the correct laboratory certificate of waiver for your testing. Complete lab credentialing for clinics with all payers. CLIA medical billing requires proper certification linked to enrollment.
FAQs
What is CLIA certification?
CLIA certification authorizes facilities to perform laboratory testing. It’s required for all human specimen testing. This includes simple tests like rapid strep and COVID tests.
How long does the CMS-116 application take?
The CMS-116 application process takes 30 to 90 days. Submit to your state health department, not CMS directly. For waived certificates, no inspection occurs.
What is PEMS, and why does it matter?
PEMS is the Provider Enrollment Management System. It’s Medicare’s electronic verification database. You must enter your CLIA number in PEMS.
When is the June hard-edit deadline?
The exact date varies, but it occurs in June 2026. After this date, automatic claim denials begin. There’s no grace period.
Can I bill for tests without CLIA?
No, you cannot bill for any lab test without a valid CLIA certification. All payers require CLIA for lab reimbursement.





